Musculoskeletal pain is a condition that may arise from a variety of circumstances ranging from injury to genetic disorders, and can drastically reduce a person's mobility and quality of life. Pharmacologic treatments include both non-narcotic and narcotic options, although these may result in unwanted side effects and drug interactions. Pulsed electromagnetic field (PEMF) therapy holds promise as an exciting, effective, non-pharmacologic alternative to traditional treatment regimens. In this article, IDTechEx will detail the recent approval, advantages, and potential additional applications of PEMF technology.
Impact and traditional treatment of musculoskeletal pain
The prevalence of musculoskeletal pain is on average 30%, while that of chronic widespread pain (typically lasting more than 3 months) has been reported to be as high as 24%. Non-narcotic treatments typically include NSAIDs, antidepressants, anticonvulsants, and capsaicin. Although the use of non-narcotic therapies can sometimes be ineffective, and as many as 5% of US adults, for example, are treated with narcotic opioids. Both non-narcotic and narcotic treatments, however, have the potential to interact with medications patients may already be taking for other diseases. Furthermore, opioids are highly addictive and can be dangerous, responsible for a roughly sevenfold increase in methadone-related deaths in 2009 alone.
PEMF approval and efficacy
PEMF technology has been FDA approved for bone-related indications since 1979, but just recently gained approval for the treatment of musculoskeletal pain in February of 2017 with BioElectronics' ActiPatch®. This particular technology is currently approved for over the counter (OTC) use in both heel plantar fasciitis and knee osteoarthritis. However, in a 2015 Pain Management study that garnered 5002 responding participants, this technology was found to reduce general musculoskeletal pain stemming from a variety of conditions by an average of 57%. Emitting a pulse 1000 times per second, the ActiPatch® reduces the perception of pain by altering afferent neuronal pathways that transmit these signals to the brain. This same pulsing feature also prevents the patient from adapting to the therapy, likely rendering it effective over a longer period of use.
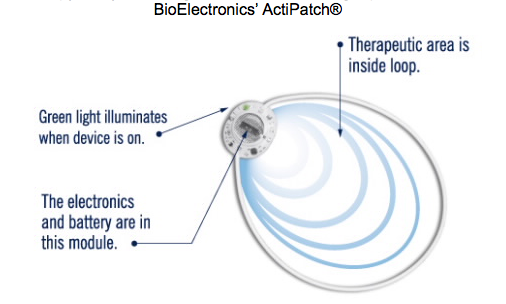
Image source: BioElectronics
PEMF as an innovative, and competitive, alternative
As a "drug-free" treatment for musculoskeletal pain, PEMF technology could circumvent those drug interaction and addiction issues associated with traditional pharmacologic management methods. Furthermore, the devices such as the ActiPatch® are proposed to last several months. Retailing for $30, the average cost to a patient per month could be expected to be at most $10. Such a figure is impressive given the stark contrast to traditional treatment costs. Published estimates from the Journal of Managed Care & Pharmacy reveal monthly patient costs for NSAIDs to be as high as $104, and those for opioids over twice as much at $251 per month.
PEMF future applications
The non-invasive, non-pharmacologic nature of PEMF technology makes it an attractive candidate for the treatment of other pain-related conditions. Two potential examples may include fibromyalgia or 'phantom limb' pain. Fibromyalgia (FM) is a disease of diffuse, chronic pain with unclear causes. It is characteristically difficult to treat, with only 3 FDA approved medications indicated for this purpose. Phantom limb is a condition where a patient continues to feel an often painful sensation arising from a previously amputated body part. The condition, with a debated etiology, also suffers from a lack of effective therapeutic options. Affecting approximately 10 million and 185,000 people respectively in the United States alone, FM and phantom limb represent two large patient populations that stand to greatly benefit from the demonstrated therapeutic efficacy of PEMF technology.
Top image: East Bay Wellness


